Last Updated on July 7, 2024 by Max
Introduction
For over 75 years, it has been accepted that higher serum testosterone increases the risk of developing both new and rapid growth of latent prostate cancer. How did it happen that testosterone, being a hormone controlling the development of the prostate early in life, becomes a destruction and degeneration factor of the prostate in old age? What is the price of the risk of testosterone therapy, and does it prove the expected benefits?
There is considerable evidence that androgen deprivation therapy noticeably depresses the development of prostate cancer in humans and prostate cell line experimental systems. However, there is no clear evidence that an increase in endogenous testosterone levels contributes to the development of prostate cancer in men.
Let’s study the issue carefully, analyzing the pros and cons of available evidence-based data.
Who Needs Testosterone Therapy?
In adult men aged 20 to 40 years, the average level of testosterone has been accepted as 630 ng/dL, with the normal rates ranging from 264 to 916 ng/dL With age, especially after 50, the levels of testosterone in men’s blood decline noticeably, leading to a bunch of negative consequences, often changing our habitual lifestyle, affecting our sexual life. Some men take it for granted and resign themselves to their changing health status. However, many men do not want to come to terms with this and are ready to break their habits and change their lifestyle and diet to counteract the onset of the changes that testosterone deficiency entails. Usually, these are healthy men who are ready to make some efforts and sacrifice their time to prolong the stunning fullness of life and delay the onset of biological oldness.
A subset of men who need testosterone correction also includes people who suffer from symptoms of pathologically small testicles. The secondary reduction of the test size is usually associated with a variety of other health conditions, including hypertension, diabetes, and obesity, and is observed in more than 2.5 million US men over 40 years of age. Experts predict a growth trend for this category of men in the future.
But here, an unexpected threat in the face of prostate cancer awaits them. Ironically, at the same age, as the background of blood testosterone levels decreases man’s prostate grows enough to cause numerous bothersome symptoms. Why it happens, you can read here. The growth of the prostate tissue is under the control of testosterone. And here the question arises, whether our attempts to keep up the optimal level of testosterone in the blood will lead to malignant degeneration of prostate tissue.
The third group of men includes those with a prior prostate cancer history.
How does testosterone work?
Testosterone is the key hormone controlling the development of male reproductive organs such as the prostate and testes. The principal sites of testosterone secretion are testicles in males and, to a lesser extent, ovaries, and adrenal glands in females. In women, this steroid is produced as much as 7-8 times less than in men, though it plays an essential role in the setting of their libido, ovarian function, and bone strength.
The precursor of testosterone in the body is cholesterol, but it doesn’t mean that you can elevate your testosterone level by eating a high-cholesterol diet. The importance of testosterone for our health is too high to be able to change it so easily. There is a gentle mechanism by which our brain controls the blood levels of testosterone. We must know how this mechanism runs to take care of it if we want it to work smoothly throughout our lives.
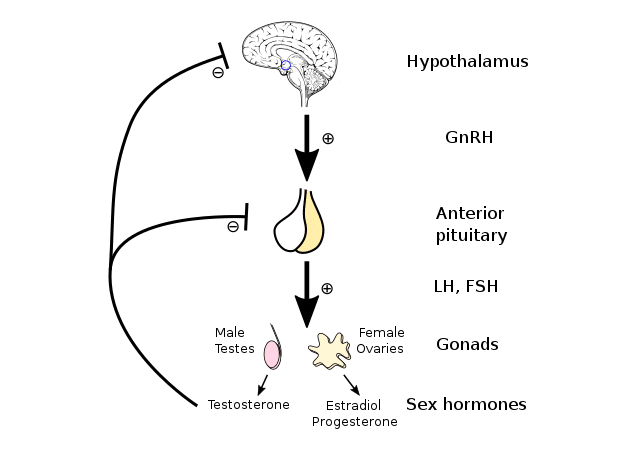
At the very base of the brain, we have a small area called the hypothalamus. While it’s very small, it plays a crucial role in connecting nervous and hormonal systems, transforming the signals from the brain into physiological reactions via releasing hormones in multiple sites throughout the body.
When testosterone levels in the blood are low, the hypothalamus releases a special substance called gonadotropin-releasing hormone (GnRH) into the pituitary gland. The pituitary gland is a tiny gland no larger than a pea and is directly connected to the hypothalamus. GnRH stimulates the pituitary to release follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which in turn stimulate the testes to synthesize testosterone.
By Artoria2e5 – Own work, CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=81460023
On the contrary, increasing levels of serum testosterone depress the synthesis of GnRH and FSH/LH in the hypothalamus and pituitary, respectively, which in turn slow down the production of testosterone in the testicles. The coordinated work of the hypothalamic-pituitary-testicular axis is very important for maintaining the blood testosterone at a healthy optimal level.
How Does Testosterone Is Related to Other Sex Hormones And Prostate Physiology?
5α-dihydrotestosterone (5α-DHT) and estradiol are two direct derivatives of testosterone, biological functions of which are capable of enhancing the development of masculinity (5α-DHT) or femininity (estradiol ) of a male individual.
5α-DHT mediates the development of the external genitalia, urethra, and prostate. With the onset of puberty, sex hormones contribute to the rapid growth of an immature prostate, increasing its size ten times.
It has been well established the principal role of 5α-DHT is in promoting the continued growth of the adult prostate, leading to BPH.
An enzyme 5α-reductase converts testosterone into 5α-DHT, a more potent hormone, in the liver, and male reproductive organs, such as the prostate, seminal vesicles, and epididymis. Moreover, the expression of 5α-reductase is detected in hair follicles, the brain, and the skin. While the circulating levels of 5α-DHT are about 10% of those of testosterone, in reproductive organs with high 5α-reductase activity, it amounts to as much as 90%. It has been calculated that in these organs, the effect of testosterone is potentiated 2- to 3-fold due to a higher affinity of androgen receptors to 5α-DHT than to testosterone. When we use 5α-reductase inhibitors, either to alleviate the symptoms of BPH or to slow down hair loss, we must understand that in this way, we lower the concentration of active androgen to relieve symptoms temporarily, but we don’t solve the underlying problem.
Another enzyme, aromatase, converts about 0,3% testosterone into estradiol. Aromatase activity is high in the liver, adipose tissue, brain, and bone. The high activity of aromatase in adipose tissue explains the low libido and feminine shapes in obese men.
The Effects of Testosterone Treatment on Prostate Health
In 1941, Huggins and Hodges, for the first time, reported the beneficial effects of castration on the regression of metastatic prostate cancer, for which the authors were awarded the Nobel Prize. Later, the results of these studies were repeatedly confirmed. Fowler and Whitmore (1981) reported that 45 of 52 men with metastatic prostate cancer treated with testosterone resulted in unfavorable outcomes. As in the previous study, almost all of the patients had already undergone androgen deprivation treatment (ADT) when they received testosterone. Surprisingly, no unfavorable results were reported for three among four men who did not get ADT and who received testosterone injections for up to a year.
The same results were reported by Prout and Brewer (1967), who observed rapid progression in prostate cancer in half of the men who had been previously castrated when given testosterone. However, no adverse results were found in patients who received testosterone but were not castrated beforehand.
These results suggested that testosterone therapy did indeed stimulate prostate cancer progression in castrated or androgen-deprived men, but not in men with untouched testosterone status. Clinically, ADT remains a mainstay in advanced prostate cancer treatment.
Along with the results of the adverse effect of exogenous testosterone on the progression of prostate cancer, there has been accumulating conflicting clinical data on the role of testosterone in prostate cancer pathogenesis. These data show no clear elevation in prostate cancer rates following testosterone administration in normal men or men at high risk for prostate cancer, no connection of prostate cancer risk with testosterone levels, and no reduced risk of prostate cancer in patients with low testosterone.
In 2012 Muller et al. analyzed 3242 men between the ages of 50 and 75 years who all had at least one prior negative prostate biopsy. The researchers found no association between testosterone or DHT levels with prostate cancer risk.
Similarly, Roddam and colleagues identified no association between prostate cancer and serum testosterone after a large meta-analyze, including data from 3886 prostate cancer patients and 6438 controls.
A list of research denying the relation between the degree of prostate cancer risk and the high levels of blood androgens has been replenishing every year. On the contrary, clinical studies have mainly correlated lower testosterone levels with prostate cancer hardness.
Currently, available data do not suggest an increased risk of prostate cancer in men undergoing testosterone therapy. Haider and colleagues reported the results of 1023 hypogonadal patients who were treated with exogenous testosterone and followed up for five years. The incidence of prostate cancer was lower in testosterone-treated populations than in controls. Likewise, in multiple smaller studies of men at increased risk of prostate cancer, testosterone treatment proved to be safe with respect to prostate cancer risk.
The link between testosterone therapy and a higher prostate cancer risk is unclear. However, a study published online on June 22, 2018, by PLOS One found no connection between testosterone therapy and aggressive prostate cancer among more than 58,000 men ages 40 to 89.
How Does Testosterone Stimulate Prostate Growth?
So we have an apparent paradox in which castration or ADT makes prostate cancer revert, yet higher testosterone levels fail to induce prostate cancer to grow. To explain and reconcile seemingly contradictory scientific and clinical results, a saturation model was proposed, according to which maximal stimulation of prostate cancer is reached at relatively low levels of testosterone. Prostate tissue growth is extremely sensitive to changes at very low androgen concentrations but becomes indifferent to that at higher levels.
The saturation point has been approximately calculated to be 250 ng/dl of testosterone. Decreasing serum testosterone concentrations below this point will cause a reduction in prostate size for men with benign or malignant prostate tissue. Increasing testosterone levels anywhere up to the saturation point will cause prostate tissue growth. However, the further rise of serum testosterone above this point will result in limited or no additional growth of prostate tissue.
The saturation model is consistent with the observation that androgens exert their effects by making an androgen-androgen receptor (AR) complex in the cells. When we add some exogenous testosterone into the blood, it binds with all ARs available in the prostate tissue until saturation. Once the maximal androgen-AR binding is reached, the presence of extra androgen performs a limited additional effect.
Conclusions
In conclusion, the available evidence-based data strongly suggests that testosterone therapy is safe for prostate health. Since the population of men symptomatic of testosterone deficiency is largely the same as the population of men at risk for prostate cancer, it is strongly suggested that men undergoing testosterone therapy undergo regular screenings for prostate cancer.
Further research is required to determine the safety of testosterone therapy in men with prostate cancer.
Until now, despite the numerous clinical data and research results with small numbers of patients, there are no wide-scale randomized controlled trials on the effect of testosterone therapy in the men population at risk for prostate cancer. Therefore, testosterone therapy may only be offered to select men who are strictly monitored and well-informed about the potential risks and benefits.

References
- Editor(s): Jones, T.H. (Barnsley/Sheffield). Advances in the Management of Testosterone Deficiency. ISBN: 978-3-8055-8622-1
- Abraham Morgentaler. Testosterone and Prostate Cancer: An Historical Perspective on a Modern Myth. Eur Urol. 2006 Nov;50(5):935-9. doi: 10.1016/j.eururo.2006.06.034. Epub 2006 Jul 27.
- Jason E. Michaud, Kevin L. Billups, and Alan W. Partin. Testosterone and prostate cancer: an evidence-based review of pathogenesis and oncologic risk. Ther Adv Urol. 2015 Dec; 7(6): 378–387.
- Abraham Morgentaler and William P Conners III. Testosterone therapy in men with prostate cancer: literature review, clinical experience, and recommendations. Asian J Androl. 2015 Mar-Apr; 17(2): 206–211.
- Huggins C, Hodges CV Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. 1941. J Urol. 2002 Jul; 168(1):9-12.
- Fowler JE Jr, Whitmore WF Jr. The response of metastatic adenocarcinoma of the prostate to exogenous testosterone. J Urol. 1981 Sep; 126(3):372-5.
- Prout GR Jr, Brewer WR. Response of men with advanced prostatic carcinoma to exogenous administration of testosterone. Cancer. 1967 Nov; 20(11):1871-8.
- Muller R., Gerber L., Moreira D., Andriole G., Castro-Santamaria R., Freedland S. (2012) Serum testosterone and dihydrotestosterone and prostate cancer risk in the placebo arm of the reduction by dutasteride of prostate cancer events trial. Eur Urol 62: 757–764.
- Haider A, Zitzmann M, Doros G, Isbarn H, Hammerer P, Yassin A Incidence of prostate cancer in hypogonadal men receiving testosterone therapy: observations from 5-year median followup of 3 registries. J Urol. 2015 Jan; 193(1):80-6.







